Who we are:
Nutek Bravo is a ISO-certified sterilization, science, and engineering company, based in the San Francisco Bay Area. We provide reliable, convenient, and accessible irradiation services using Electron Beam processing. This sterilization technology is a cost-efficient and modern alternative to other modalities, such as Gamma or Ethylene Oxide.
The Nutek Bravo facility in Hayward, California performs batch processing, process validation, and product development for our clients.
The Nutek Bravo facility in Hayward, California performs batch processing, process validation, and product development for our clients.
Irradiate your products with us in 3-steps:
We are happy to take all inquiries regarding our services at Nutek Bravo. If you are interested in processing product at Nutek Bravo, then use these three steps:
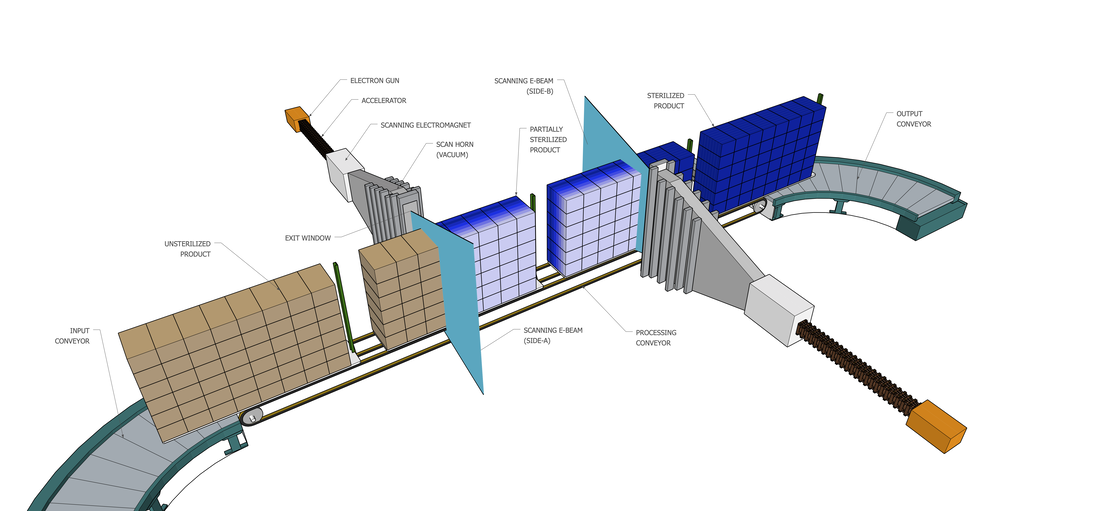
Our E-beam systems:Electron beam (E-beam) processing uses high energy accelerators at high power to provide dose penetration throughout each product the beam hits. Products are placed on our carriers and proceed through the opposing, scanning E-beams at a controlled and set speed to deliver a validated dose.
|